Not Just a Pretty Face: Why Museums Need Study Skins
Warning: This post contains information and images pertaining to the preparation of dead animals via taxidermy, and may not be appropriate for readers who find this topic disturbing. (The rest of us may find it helpful as a holiday season appetite suppressant.)
Museum of natural history: What comes to mind when you think of these words? Dioramas, taxidermy, giant articulated skeletons of charismatic megafauna…and if you’re from around here, a rattlesnake that shakes its rattle when you push the big red button. But our exhibits are just the tip of the iceberg. Indeed, most museums have collections behind the scenes that extend far beyond the selection of specimens on display. In our case, the Collections & Research Center (CRC) houses over 3.5 million specimens and artifacts, as we never tire of bragging. Although our exhibits team and education staff draw on those collections to inform the public, most of the time the specimens in the CRC are shared with a different audience: visiting scientists who come to study them to advance their research. Yet the CRC specimens are stored within a modest 12,937 square feet of space, while our halls and outdoor exhibits sprawl across a far larger campus. How do they fit all those specimens in there? In the Invertebrate Zoology Department, it helps that many of the treasures are tiny, from microshells to microscopic insects. The Vertebrate Zoology Department has no such luck. They’re the ones who have to figure out where to put the whale skulls.

A young visitor to VZ sizes up some of the whale skulls in the collection.
Curators of Vertebrate Zoology Krista Fahy, Ph.D. and Paul Collins, M.A. have a secret up their sleeve (or, if you will, up their drawers): study skins. These are specimens preserved using a style of taxidermy that differs from the picturesque mounts you see in our dioramas. Dr. Fahy explains: “If you were going to do a taxidermy specimen to go on display for the public, it would be in a lifelike pose, it would have glass eyes…study skins are flat on their backs so they don’t roll around, take up the minimum amount of space, and the more you can get in a drawer, the better.”

Guests who get a behind-the-scenes look at collections of study skins are sometimes put off by the distinctly dead appearance of these specimens, with their flashes of white cotton at the eyes and eerie rigidity. But for curators and researchers, they’re perfect; they preserve the morphology—the external characteristics—of a specimen in the most efficient way. When it comes to birds, researchers consult a specimen to note characteristics of “the feathers, the color and length of the beak, any of the external measurements,” Fahy reports. The skins of mammals feature furs instead of feathers, but the concept of preserving morphology is the same. Studying these features helps scientists determine what distinguishes a species, how that species changes over time, and how it relates to its fellows on the tree of life.
Study skins are part of an old tradition, but the methods used to learn from them are constantly changing. In turn, this has changed how they are prepared. During the Victorian period, Fahy says, scientists were mainly interested in “documenting the diversity of birds on the planet, [so] the skin was enough. Now we’re looking at different levels of analyses.” One consequence of this is that we now prepare study skins alongside a whole suite of other resources. It’s the scientific equivalent of the culinary nose-to-tail ethos: we preserve just about everything that can be of use, and take extensive notes about the parts that aren’t preserved (such as stomach contents, fat stores, and reproductive organs). In addition to the study skin, an animal can yield a skeleton that can be independently examined (though the bones in a bird’s feet and parts of its head remain part of the study skin to better preserve its morphology). Samples of organ tissue can be frozen for later analysis, yielding up a wide range of information about genetics, environment, and pathology.

Dr. Fahy shows off a study skin during a recent Leadership Circles tour of VZ.
I asked Fahy what researchers can learn from study skins, and we determined it was a lot easier to say what they can’t learn. “Getting a song from a dead bird is difficult,” she admitted, “but the questions are unlimited.” When an interested researcher appears on the scene, “we can get back and look at genetics, contaminants, [and in future] all sorts of stuff...We don’t even know what questions can be answered, because we haven’t yet thought of asking them.”
If this talk of preparing and preserving specimens with undreamed-of technology in mind sound like futurist fantasy talk from someone waiting to receive a personal jetpack, consider that it’s already happened. With the advent of methods that allowed scientists to extract and analyze DNA, museum curators discovered that some of their specimens were suddenly much more information-rich. Other specimens were found to be less useful because the methods used to preserve them degraded the DNA they once contained. Alcohol and formalin—fluids used to preserve specimens in jars—can damage DNA, so now curators may choose to freeze material they would have once prepared this way. In fifty years, it’s likely that new methods to extract yet more information from these specimens will have been developed, “so our game is to be guessing what kind of techniques might come next, and just making sure we collect as much as we can, as responsibly as we can, with the best amount of data, and keep it well preserved,” says Fahy.

Tissue samples from the sub-70° C freezer
As a consequence, the freezer in Vertebrate Zoology is full of curious odds and ends, like a giant plug of earwax from a whale’s ear, which eventually yielded an amazing amount of information through undreamed-of methods. When Paul Collins collected the plug with expert assistance from Dr. Frances Gulland of the Marine Mammal Center—a heroic feat, considering this nasty prize was located deep in the ear canal of a massive, slippery, decaying whale carcass—he was following his curatorial instincts. Sure, we all have an inner voice that says, “Hmmm, this might come in handy someday,” but trained biologists and curators have educated that voice to the point that it says things like, “Well, this is a rare chance to stash some whale earwax.” Collins had no way of knowing the specific use to which this specimen would be put, but in 2007, he stored it in the freezer to keep the wax from melting.
The freshly-extracted plug in a photo that gives a sense of the difficulties of obtaining it. Truly, a plug of whale earwax in the hand is worth two in the ear canals.
In 2013, researchers who made use of the specimen published a paper on their use of novel methods to analyze the hormones and contaminants in the plug to learn about the whale’s development, stress levels, and exposure to pollutants across its life. Since earwax accumulates in the plug over time, they were able to see the “lifetime profile” of how these substances were laid down in the wax—like rings in a tree—to a resolution of six months. This year, further work by the same researchers made the news again, as they published data tracking stress hormone levels in many earwax plugs, dating as far back as the 1870s. The resulting graph correlates pretty closely with the history of stressful times to be a whale, marking peaks of whaling industry activity and underwater noise.
But enough about whale earwax! (Words we confidently claim you will never hear anywhere else…) The point is that since the most unassuming specimen might turn out to be a future treasure trove of data, the preparation process is approached with great care. “It’s almost like an autopsy on a human,” says Fahy. “We’re trying to get all those fine details, because the preparator’s the only one who’s ever going to see the guts.” So it’s appropriate that the main preparator at SBMNH is Curatorial Assistant Julia Schorr, M.A., whose background in anthropology included an internship with the coroner’s office in Denver, CO. Schorr finds preparing study skins less intense than performing autopsies, but her stint working with human bodies ensured that she has the requisite guts for the task. It also takes fine motor skills, great attention to detail, and—when done right—a real artistic sensibility.

Schorr prepares to work on a juvenile Red-tailed Hawk.
“Most of the really good bird preparators have an artistic background,” says Fahy. “There’s beauty to it, to make sure that each feather lays down in the right track. You can see people who prepare who are just trying to get the bulk of [their specimens] done and in the drawers, and there’s people who really take pride in what the specimens look like.” Schorr is one of the latter. “You see how the researchers come in and go, ‘Ooohhh, who did this bird? This looks good.’ I think there’s a pride in that, rather than turning out gruesome little mangled skins.”

A pair of Scarlet Tanager study skins. One was probably prepared with greater care.
As a preparator, Schorr sees the good, the bad, and the ugly of what makes it to the departmental fridge. Some specimens come from other institutions, some are collected by Museum staff (often tipped off by roadkill-alert members of the public), and some are brought to us by concerned citizens. Birds that ran into windows and never recovered, animals poisoned (directly or indirectly), animals killed by conspecifics (other animals of the same species) in territorial conflicts…what they all have in common is that they’re dead, and are beginning to decompose. “If they’re not super-fresh,” Schorr says, “the skin starts to tear a lot more easily...You just have to be a lot more delicate with them and hope that they hold together enough that you don’t waste a bunch of time trying to get a skin and then have to scrap it.” A violent end makes her job harder, too: “It’s always challenging if they’re damaged on the inside from injury, working around broken bones makes it harder.” It’s a testament to how well-suited Schorr is for this job that—when asked about the challenges of the work—she didn’t even mention the smell. For an outsider in the VZ side lab (where Schorr does much of her work), the smell is the most striking feature of a workplace with many striking features.

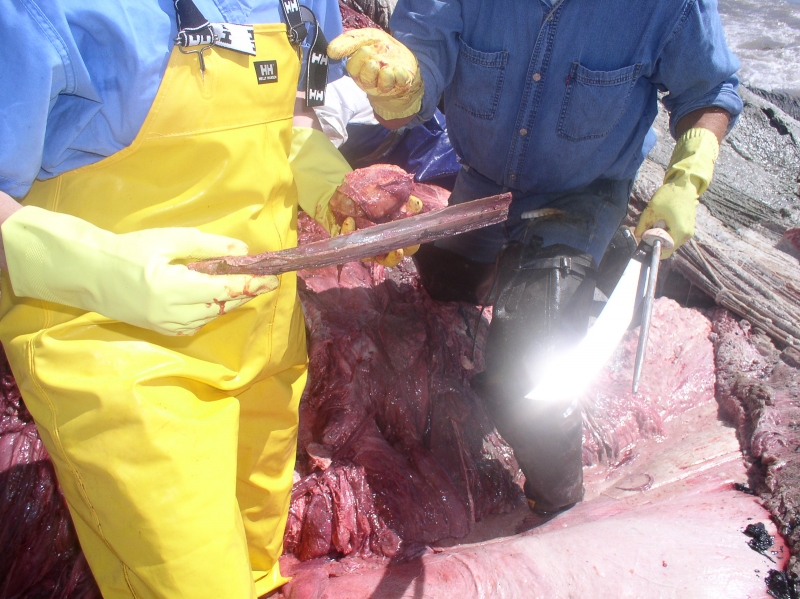
The side lab is a long, shotgun-shack space with a garage door at one end (essential for loading mammoth skulls and other large, awkward specimens) and a screen door—often propped open for essential ventilation—at the other. Like other multipurpose spaces around the Museum, it’s a cabinet of curiosities in itself. Come in on the right day, and you’ll find a defrosting Bigeye Thresher Shark atop the necropsy table or a rack of fossilized sea cow ribs in the middle of the room. There’s plenty of specialized equipment in here, including a purpose-built gentle tumble dryer for removing moisture from freshly-washed specimens, and a brush dremel for scraping fat remnants from the insides of skins. Hungry dermestid beetle larvae are hard at work in a tank, disturbing skeleton bundles hang suspended in a pie-cooling cabinet, and a double row of jars next to the sink are full of cloudy fluid and fuzzy-looking bones (more on all that later). A seven-foot Japanese whale flensing knife is discreetly tucked behind a baker’s rack full of bins. There’s a massive freezer which constantly hums, as well as fans that buzz and a stereo that amplifies podcasts Schorr plays from her phone (true crime, of course). And then there’s that smell.
The smell is partly the unwholesomely clean odors of chemicals (including the abovementioned alcohol and formalin) that pervades the entire CRC. Facilities crew and CRC staff alike keep the facility spotless, to safeguard everyone’s health and protect samples from cross-contamination. But the rest of the heady atmosphere is supplied by the slow process of decomposition. That natural process has been arrested (often for years) by a waiting period in the freezer, since we have a backlog of specimens to preserve. The goal of preparation is to stave off that process much, much longer, so the specimen remains useful to researchers far into the future. (Entomologists just rejoiced over the discovery of mummified beetles preserved by ancient Egyptians in 2,500 BC, so we can dream big.)
Recently Schorr prepared a study skin of unusual size: a California Condor. It’s not every day we see a condor specimen around here. These massive birds were nearly driven to extinction by over-collection and lead poisoning (they consume fragments of lead-based ammunition when they eat their natural diet of carrion). As they are listed as critically endangered today, collecting is no longer permitted, but lead poisoning remains a threat. By 1987, all the 27 known living California Condors had been brought in from the wild to participate in a captive breeding program, and today their descendants—now numbering in the low hundreds—still rely on human friends for a lot of assistance, including monitoring their health for problems with lead. If you’re a hunter or rancher, you can help condors (and other carrion-eating wildlife) by using lead alternatives and encouraging your peers to follow suit.

Schorr assessing the condor prior to preparation. The orange bit in the middle is what remained of the carcass post-necropsy.
Anyone who’s been lucky enough to see a condor in flight knows why they’re worth protecting. In a photo, an illustration, or on a TV screen, they sometimes feel like big Turkey Vultures, but in person, when they soar, there’s no mistaking their graceful glide for the teetering flight of their lesser cousins. Even in death, they’re pretty damn inspirational. The bird Schorr worked with was a juvenile whose head was still dark and sparsely downy, and the plumage on the undersides of her wings wasn’t yet the distinctive bold white. After an accidental death, she had been thoroughly necropsied (the non-human equivalent of autopsied) many years ago by the source institution, a captive breeding facility run by the Peregrine Fund, one of the groups cooperating in the effort to save the condors.
Eventually, the specimen made its way here, largely through the influence of Jan Hamber, the archivist of the California Condor Archives (a resource housed here at SBMNH). Hamber has deep connections to the condor world because she’s devoted much of her life to the work of studying and protecting the species. And as someone whose connection to SBMNH began with work on specimens going into exhibits, Hamber even has personal feelings about the distinctive smell of museum work. In a 1959 letter recounting her first days here, she wrote: “The minute I walked up the stairs to the lab area and smelled the faint aroma of formaldehyde, I felt at home.”

Jan Hamber of the California Condor Archives
Fahy clearly appreciates Hamber’s advocacy for getting birds out of institutional freezers and into museum collections. “She understands the value of getting the birds into a publicly accessible research institution,” said Fahy, noting the importance of having specimens—especially specimens from rare species—distributed across many collections. When Brazil’s National Museum was consumed by flames this September, the loss to science and culture was devastating. One of the lessons reinforced by that tragedy: don’t keep all your condor eggs in one basket. So we’ll have our share of California Condors, and no more.
Although we have a few condors already in our collections, this was Schorr’s first time working on one, and she was “really excited to get to work [on it]. Not many people get to see them, let alone touch them.” Schorr wasn’t always a bird freak. “Before I started working here, I was kind of neutral on birds,” she admitted. “Some of them are pretty, some of them are brown and boring. Once I started working here and getting up close, you can see how pretty they all are. You see the range of sizes and colors. Getting to see this huge condor was super fun.”
I asked Schorr if she felt a lot of pressure working on the condor. “I wouldn’t say it was just another day at the office,” she admitted. “It was a special day. Definitely putting in a little bit more effort than I would into a little Hermit Thrush or something I get all the time.” First, there were routine steps: assessing the condition of the defrosted specimen, confirming ID, weighing it and taking various other measurements. Because the specimen is rare, Schorr took more samples than she would for something more routine. Sure, it had already been the subject of a necropsy, but whose curatorial instincts wouldn’t tell them to save some condor tidbits for the freezer? You never know when they might come in handy.

Some early stages: weighing the bird, confirming ID (a no-brainer in this case), taking tissue samples
Then things got a little gory. After taking tissue samples, Schorr had to extract what remained of the carcass from the skin. This requires a good knowledge of the animal’s anatomy. The preparator has to disarticulate some bones, cutting ligaments to do so, and then there’s a bit of body origami (or assisted yoga). Watching it is like observing a grotesque magic trick. Snip here, push there, finesse it around for a bit, and voilà, inside-out bird! Schorr makes it look easy, but it’s real work. As her former colleague Jessica Bullington told me, you have to know how much a skin is prepared to stretch. “With the bigger ones, the skin is thicker, and you can kind of manhandle it a little bit. The little tiny ones, they just rip, and then you cry.”

The feet and some of the bones in the limbs remain with the skin to preserve its morphology.
Having separated the carcass from the skin, Schorr stripped it of most of the meat, which was then discarded. The bones that still had a little flesh on them would be cleaned by our tiny assistants: the dermestid beetle larvae do the coarse work, and microscopic organisms give the skeleton a final polish. The dermestids need to be kept carefully isolated because study skins are, in Fahy’s words, “just like dried beef jerky. They’re delicious, and there’s a whole host of museum bugs that want to come and eat your collections.” Curators inspect every drawer in every cabinet on a regular schedule to make sure any outbreak could be rapidly contained. “That’s why our drawers are lined with white archival paper, so you can easily see when damage is occurring.”
The microscopic organisms are involved in a process called maceration (which refers to the breakdown of food). You might have heard the word maceration used in reference to humans chewing, but in this case, bacteria are eating up the little bits of muscle, fat, and connective tissue that are still stuck to the bones. The dirty work is done inside the spooky bone jars near the sink in the side lab. As time passes and the bacteria do their thing, the water becomes more nasty-looking, but the skeletons get cleaner. As the condor’s skeleton went through this process, the skin continued on its own journey.

Macerating specimens. An alternate title for this blog post was “The Nastiest Cooking Show in the World.”
When a bird’s already been necropsied, there’s a long incision down the underside of its body, running all the way from the vent to the mandibles. This provides the preparator with a nice big opening through which to perform the inside-out-bird maneuver, but it also means that various bodily substances (including blood) may have dribbled out and messed up the feathers and the outside of the skin. So the skin was treated to a little bath in cold water (warm water makes the feathers fall out) and a little Woolite.

Hand wash with care; this precious item can’t be replaced.
After the bath, it had to be dried. Prior to acquiring their custom-built dryer, Paul Collins used to clean feathers with white gas. “It’s amazing I never blew myself up,” he says of the bad old days. The new dryer “has been a godsend.” After gently padding the skin with clean towels, Schorr added some extra fragments of dried corn cob to absorb moisture in the still-sticky areas inside the skin and carefully deposited the bird in the drum. After 30 minutes, she checked on it to see if it required rearranging (as any wise adult knows is necessary with certain items at the laundromat).


When it came out of the dryer, Schorr used compressed air to remove any lingering corn cob fragments and give the feathers a targeted blow-drying. Dampness can lead to mildew and decay.

At last, the skin was clean and almost totally dry. The feathers were beautiful and glossy. Collins came to inspect Schorr’s work (as he had throughout the process) and indicated his approval. He taught Schorr how to prepare specimens, and he likes to see his preparatory protégé take on new challenges like this. “I’ve been doing this over forty years,” he explains, “and you get to a point where you’ve done the same bird over and over, and Julia gets that bird the first time and she’s all excited. She’s learning new things every time she works on a new species.”

Schorr and Collins with the near-completed study skin
The skin was ready to be wrapped around a spindle of wood-fiber filler Schorr had previously prepared to emulate the size and shape of the carcass. The spindle was wound around a heavy dowel. On smaller birds, the filler material is wound around a small stick, and the stick is left to protrude from the specimen’s body because it provides a safe handle for lifting and turning fragile specimens like hummingbirds. Study skins prepared in this way are affectionately known as birdsicles. (Some small mammals assume a similar shape, with a tail made rigid by preparation—we have weaselsicles, for example—but it should be noted that we have no otterpops.) The condor was large enough to require no handle. After arranging the skin around the filler—with some cotton in the head and neck for good measure—Schorr carefully sewed up the long incision left by the scientists who performed the necropsy.

At this stage, the specimen started to resume its former shape as a three-dimensional bird. But while it retained that volume, it would lose flexibility during the next step of the process, when the skin would dry completely. Schorr folded the condor’s wings to be compact along the sides of its body, and then wrapped cheesecloth around it, pinning the cloth to a board to maintain the body’s position. In this cigar-shaped pose, the specimen went into the drying cupboard, where it remained until it was, ahem, bone dry and ready to enter the collection.

Something about the shape of birds in this position—wings folded primly at their sides—reminds one of Egyptian mummies arranged in anticipation of a glorious afterlife. Indeed, their drawers—like the tombs of kings—don’t remain shut for long. The guest book in VZ is full of entries from the visiting scientists who consult our specimens to further their research. For these animals, death may mark the end of their consciousness, but it isn’t the end of their usefulness to the world at large. Since much of the research that emerges from specimens in VZ goes on to inform conservation projects, it’s reasonable to suppose that this condor didn't die in vain, and that her body could yield secrets that may help her kind recover and repopulate their former range.

Compact storage of the condor’s skeleton in an archival box
Because the California Condor specimen is so rare, Fahy will have to act as a gatekeeper, ensuring that not just any green graduate student can access it for destructive sampling (when a part of the specimen is removed and won’t be accessible to others in the future). When the right researcher comes along—someone with a track record of publishing and sharing data, probably someone from another collections-based institution that understands the work that goes into maintaining collections—Fahy will allow them to take a little piece for lab analysis. Maybe it’ll be a tiny slice from the toe pad, from which to extract DNA; a feather or bone to analyze isotopically; or a tissue sample, to inspect for contaminants or pathology. Maybe it’ll be something we haven’t thought of yet. Whatever they glean from it, if it advances our understanding of the natural world, that makes the smell and the mess and the toil of preparation worthwhile. It makes the condor—like everything else here—something worth keeping.

Fahy and condor in the VZ collections range. (In the coming year, this room will be refitted with metal cabinets, thanks to a grant from the California Cultural and Historical Endowment.)






2 Comments
Post a CommentWhat a wonderful backstage tour of this intricate and fascinating process. Thank you for preserving the natural world for further study.
What a delightful tour of the facility and process! I especially appreciate the levity.